Playlist
Show Playlist
Hide Playlist
Alcohols – Drugs of Abuse
-
Slides DrugsOfAbuse Toxicology1.pdf
-
Download Lecture Overview
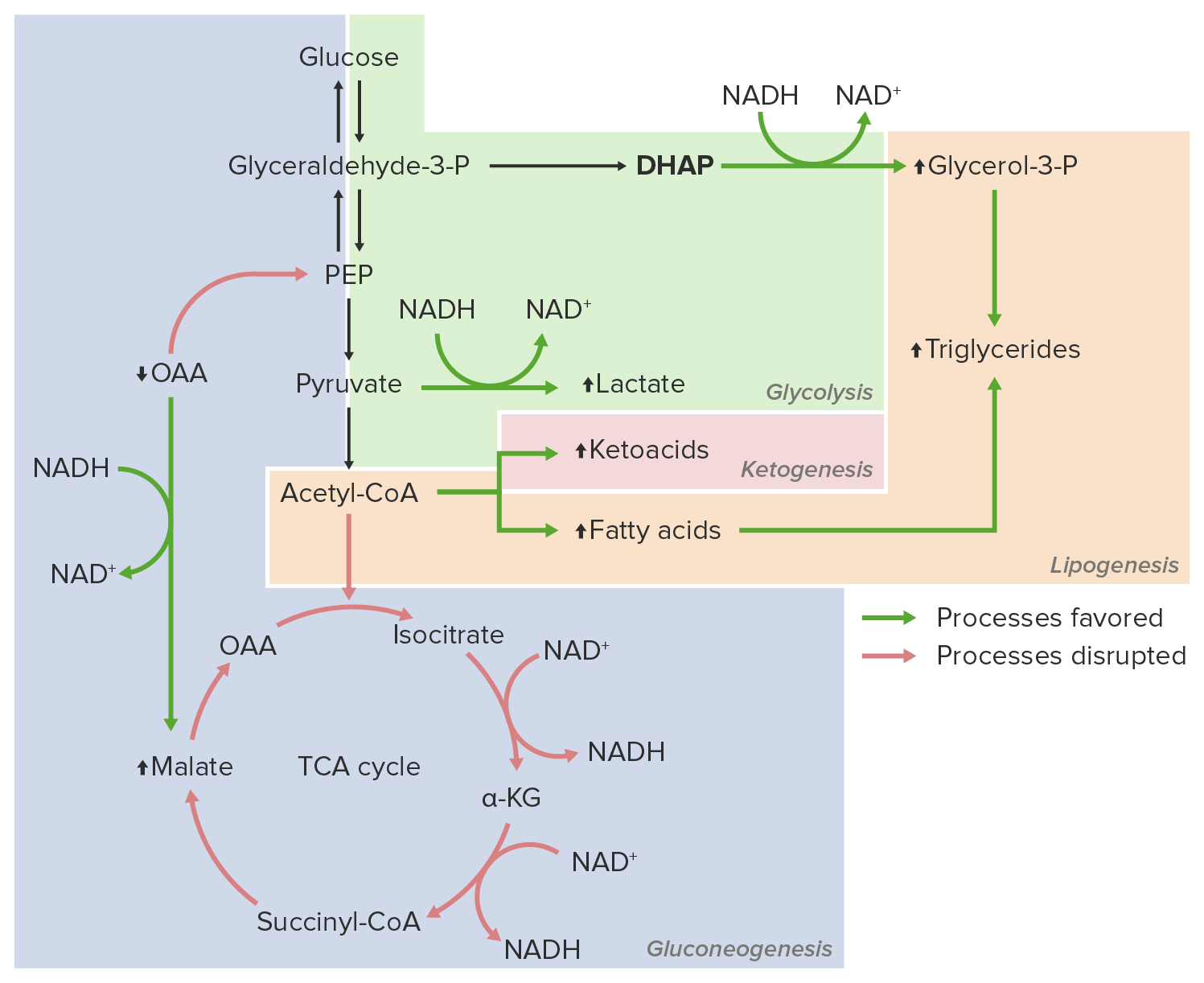
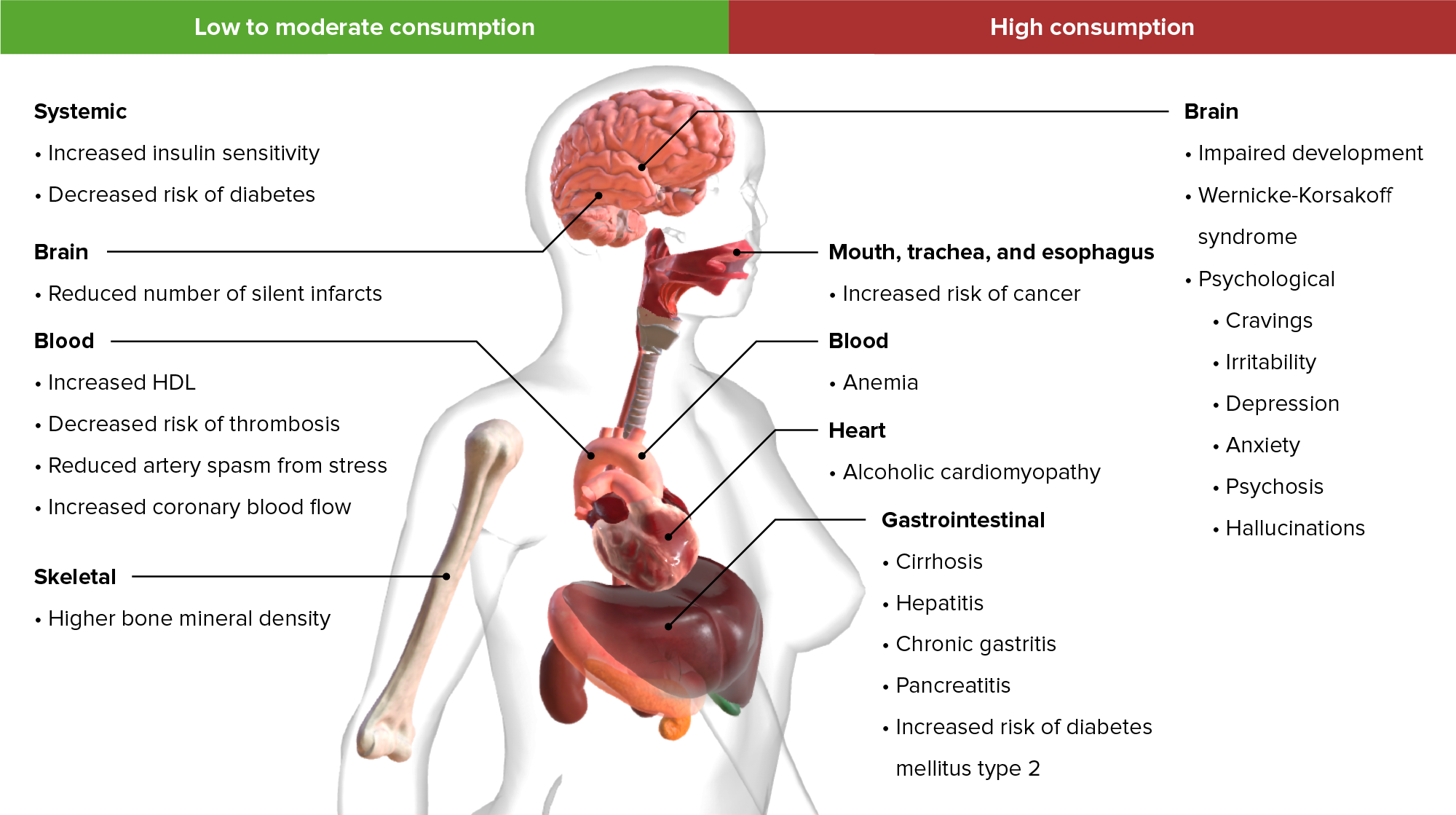
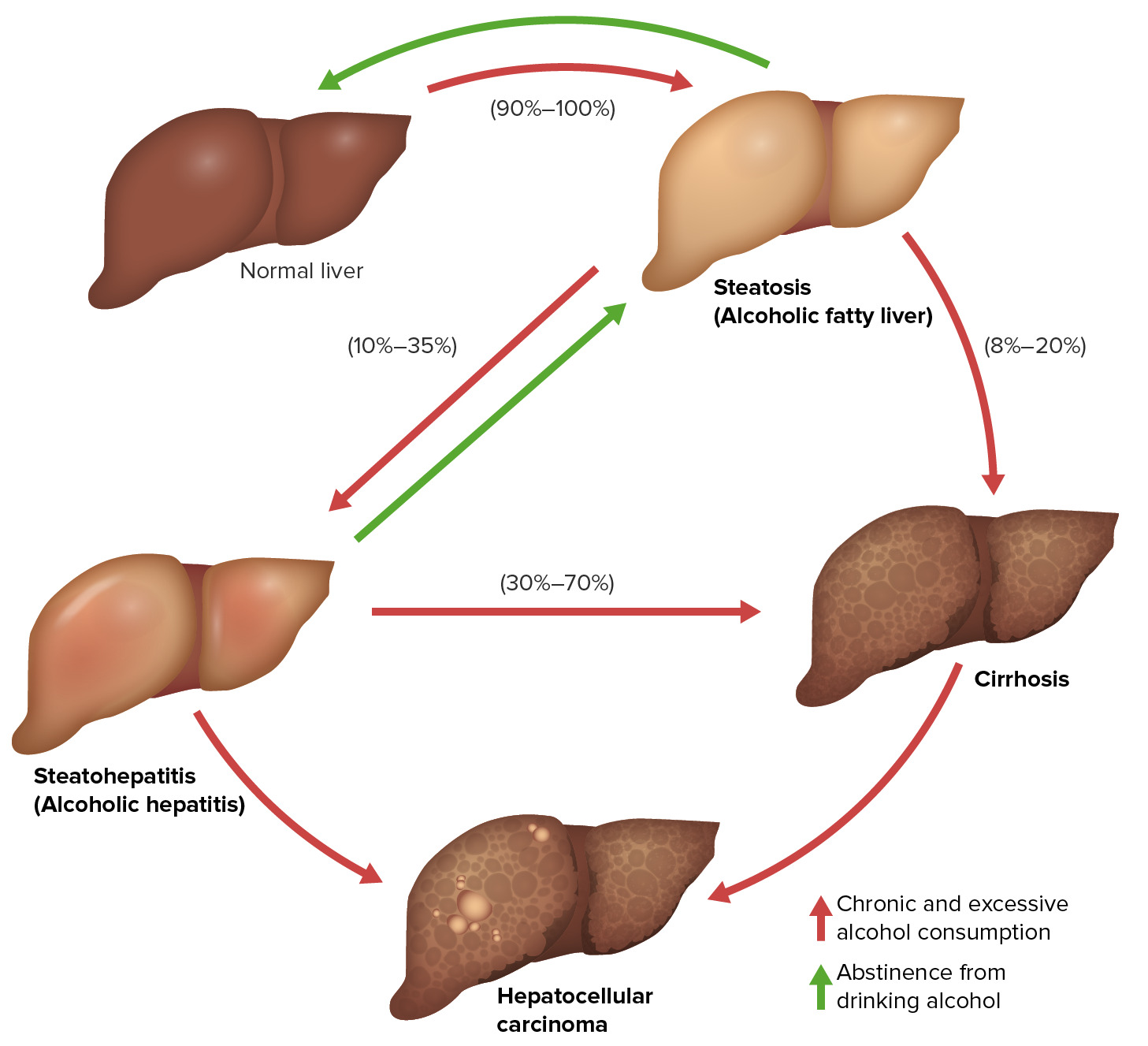
00:01 Hi, welcome to the pharmacology lectures by Lecturio. I'm Dr. Pravin Shukle and we're going to cover some toxicology today, specifically drugs of abuse. 00:11 Let's talk about alcohols. We have several clinically important alcohols and their antagonists. 00:17 In terms of alcohols, the most important ones are ethanol, methanol and ethylene glycol. 00:24 We use some drugs to treat alcohol withdrawal. We use thiamine and we also use sedative hypnotics such as diazepam. 00:32 We use alcohol dependence drugs like disulfiram and naltrexone and other medications to treat patients who are dependant upon alcohol. And finally, we treat acute methanol or ethylene glycol intoxication with drugs like ethanol or fomepizole. 00:50 In terms of how alcohol is absorbed, it's eliminated through zero order kinetics. 00:56 So if you go back to your first pharmacology lectures, we can understand what first order and zero order kinetics are. 01:02 The volume of distribution is equivalent to your body water. And it is a polar molecule, so it does cross the blood brain barrier quite easily. That's why we get intoxicated. 01:13 Ethanol is broken down by one of two systems. The first system is alcohol dehydrogenase. 01:19 It is saturatable at about 7 to 10 g/hour. And that's because you run out of NAD to power that particular enzyme. 01:29 It is more active in men than compared to women. 01:32 The second way that alcohol is broken down in the body is through the MEOS system where the microsomal ethanol oxidating system. It is active at high levels of ethanol poisoning. 01:45 And generally speaking, it can be induced because it is affected through cytochrome systems. 01:51 This contributes to acetaminophen toxicity as well and we're going to talk about that later. 01:57 Antabuse or disulfram inhibits aldehyde dehydrogenase. What ends up happening is you get a lot of accumulation of the byproducts of ethanol. You get more flushing and nausea and you accumulate acetaldehyde. 02:12 Now, one of the things that you need to know about aldehyde dehydrogenase is that it can also be inhibited by certain drugs like metronidazole, oral hypoglycemics and some cephalosporins. 02:24 And some Asians will have a deficiency of ADH, which makes them more susceptible to ethanol side effects such as flushing and nausea. 02:35 Wood alcohol or methanol is another form of alcohol that causes a lot of poisoning, and particularly in people who are moonshiners and those people make ethanol improperly. 02:46 It is also found in windshield cleaners and in some fuels. It causes direct intoxications. 02:53 So the methanol itself is intoxicating. 02:56 However, one of the problems with methanol intoxication is that the breakdown products are very very toxic. 03:02 So, formaldehyde is converted to formic acid, and causes an acidosis, causes eye damage or retinal damage, and it can cause blindness. 03:12 This is why we talk about wood alcohol blindness. 03:16 Fomepizole is a drug that inhibits alcohol dehydrogenase. So, it allows methanol itself to be excreted unchanged in the urine so you don't build up toxic levels of formaldehyde. 03:28 Ethanol is a competitive inhibitor of methanol intoxication and metabolism, so sometimes we will actually treat methanol toxicity with ethanol. 03:39 Myself have treated a number of patients who had drink wood alcohol or methanol with intravenous ethanol to the point where we actually had to intubate them and put them on ventilators to compensate for the severe intoxication from the ethanol until they can clear the methanol and then they just were allowed to wear off the ethanol on their own. 04:04 Ethylene glycol is the third most common alcohol out there. You can see that it is inhaled or absorbed through the skin. 04:11 It is often found in antifreeze. And the oxalic acid byproduct can cause severe metabolic acidosis. 04:21 Fomepizole is a drug that inhibits alcohol dehydrogenase. I had mentioned it to you earlier. 04:27 We can use it in ethylene glycol intoxication which allows the unchanged ethylene glycol to be excreted. 04:35 Once again, we can use ethanol as well as a treatment and it is a competitive inhibitor. 04:41 So, let's go to a question. A 48-year-old man was drinking heavily for 4 days. 04:47 He normally drinks less than 9 drinks per week. He is at risk for one of these five conditions. 04:58 The answer is ventricular tachycardia. So, we worried about ventricular tachycardia in this patient because it is one of the acute effects of heavy drinking. 05:08 Chronic drinkers who are heavy drinkers are at risk of Wernicke-Korsakoff syndrome or the other conditions. 05:15 But acute toxicity, ventricular tachycardia is something that we are concern about. 05:21 Let's go on to the next question. A 16-year-old boy arrives in the emergency room with confusion, ataxia and foul breath. 05:29 He is accompanied by three minors in a similar state. Blood work shows a metabolic acidosis. 05:35 Which of the following is not likely? So, let's take a look at the choices. 05:40 It could be ingestion of ethanol, of methanol, of antifreeze, of "canned heat" or "huffing" or inhaling the fumes of an unknown fuel. So, which of these is not likely? The answer is ethanol. And the reason why I want you to get this question is because you need to know that ingestion of ethanol will not cause a metabolic acidosis whereas all of the others can.
About the Lecture
The lecture Alcohols – Drugs of Abuse by Pravin Shukle, MD is from the course Toxicology. It contains the following chapters:
- Toxicology: The Alcohols
- Ethanol Metabolism
- Methanol
- Ethylene Glycol
- Question 1: The Alcohols
- Question 2: The Alcohols
Included Quiz Questions
What is a product of ethylene glycol consumption?
- Oxalic acid
- Uric acid
- Fomepizole
- Citric acid
- Acetic acid
Which drug inhibits alcohol dehydrogenase?
- Fomepizole
- Disulfiram
- Benzodiazepines
- Acamprosate
- Naltrexone
Which statement about ethanol metabolism is correct?
- Conversion of ethanol to acetaldehyde requires NAD.
- Under normal circumstances, acetaldehyde is metabolized to ethanol.
- Ethanol is primarily metabolized in the kidney.
- Conversion of ethanol to acetaldehyde requires methanol.
- Metabolism ends with the production of acetic acid.
The build-up of which byproduct of alcohol metabolism is responsible for the flushing and nausea associated with metronidazole administration after consuming ethanol?
- Acetaldehyde
- Oxalic acid
- Acetic acid
- Fomepizole
- Formaldehyde
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |