Playlist
Show Playlist
Hide Playlist
Normal Hemostasis
-
Slides Thrombus Formation Platelets.pdf
-
Download Lecture Overview
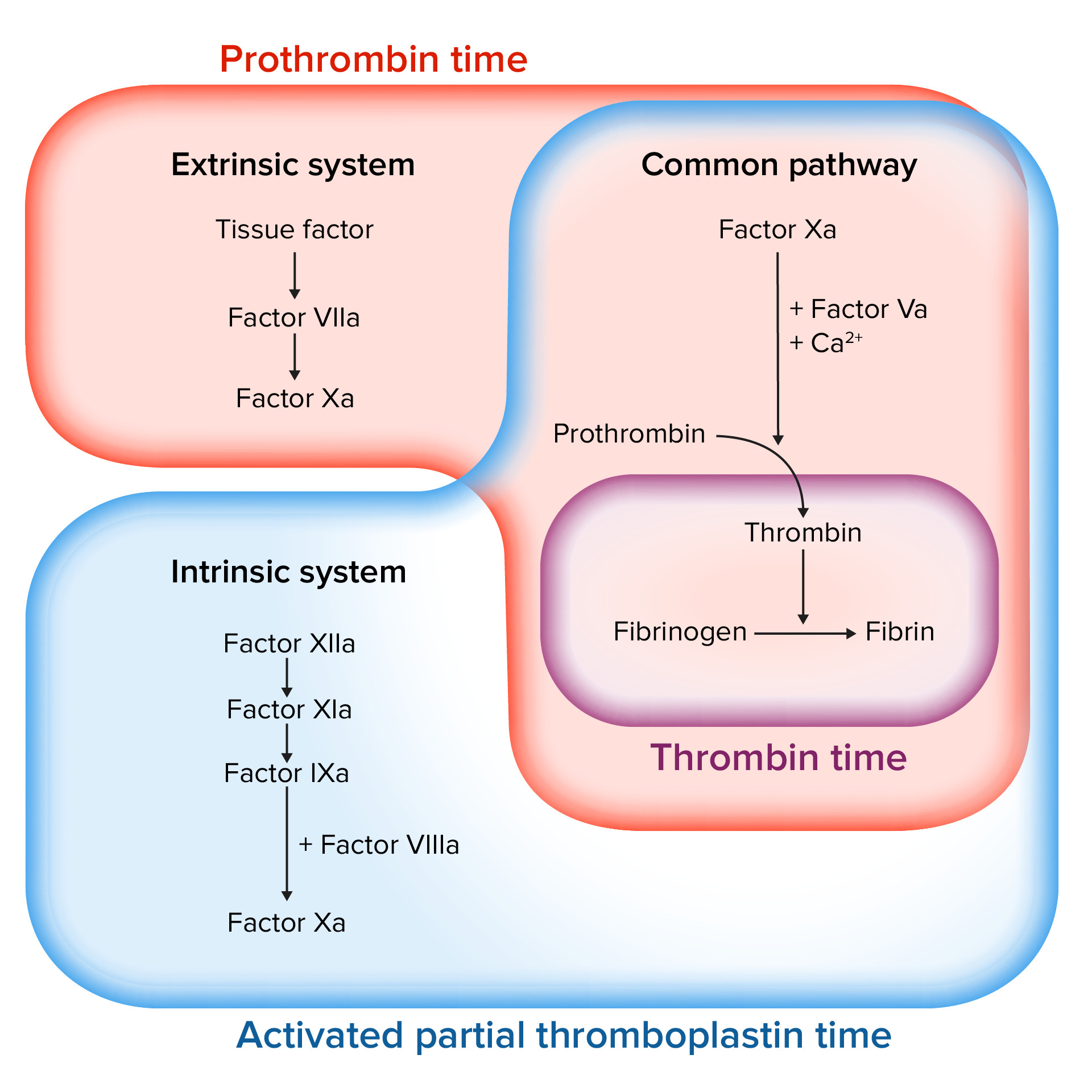
00:00 Okay, we're going to pick up and talk about the specifics now of thrombus and about clot dissolution. And this particular session is focusing on the role of platelets overall in thrombus formation. As we'll see in a minute, platelets are just one of 3 major players that will drive formation of a blood clot or thrombus. Again to remind you of the roadmap, we're going to be talking about normal hemostasis, the normal clotting that will go on and thrombus formation, but this will be part of a larger discussion overall that talks about other issues. When we talk about thrombus formation, we're going to be talking about platelets, the coagulation cascade and endothelium and they all 3 have to interact together in order to get a normal clot to form and to limit how big that clot becomes. This particular session is going to focus on platelets. Okay, let's talk about normal hemostasis. The very first step is going to be vasoconstriction as you can see at the top, but let's get you oriented to what we're looking at right now. So we're looking at a vessel and things blood is flowing from left to right across your screen. There is arterial or smooth muscle that is all the way around this artery. There is going to be a basement membrane, the basement membrane is going to be important because it has a number of constituents that are going to be important for driving thrombosis if they get exposed. Remember, normally they're not exposed because there are endothelial cells sitting on top of them as a single layer of cells keeping that blood in a liquid state. Okay, now we have a site of injury. We have lost endothelium at that site and that's going to expose the underlying basement membrane and extracellular matrix. The collagen down there is going to be one of the major drivers that will incite the formation of a thrombus. Endothelial cells though are also part of this process. They are going to release a number of factors including endothelium that's going to cause a vasoconstriction. This is going to limit flow through the area of injury. That's a good thing but it's not going to be enough to completely stop the bleeding. Also, that reflex vasoconstriction caused by the endothelial release, things like endothelin, will actually now cause diminished flow through the area that will allow platelets to begin the process of accumulating. Okay, platelets flowing through the bloodstream will, in this area of vasoconstriction, now have abnormal flow start tumbling and will be able to interact with various matrix elements sitting underneath where the endothelium has been lost. We see here one of the major factors is Von Willebrand factor (VWF). And platelets will adhere to that so you can see the platelets adhering to the underlying VWF that's been exposed and VWF has been there all along but it's been covered up by endothelial cells sitting on top of it. Once the platelets adhere, they undergo a shape change. They will also release granules to recruit additional platelets. That granule release we'll talk about in much more detail in a moment. This initial binding of platelets over the denuded endothelial surface where we've lost the endothelial cells is called the primary hemostatic plug. And again, it's the first step but it's not going to be sufficient to stop the bleeding. We're going to have to go a few more steps to make that happen. That's happening thru the granule release and the granules that are being released include adenosine diphosphate and thromboxane A2, and we'll return to those details in a moment. Those release components ADP, thromboxane A2 will lead to the secondary now recruitment of additional platelets. That's going to give us our definitive hemostatic plug. Building that definitive hemostatic plug will also activate additional factors, the coagulation cascade. So, again, endothelial cell is very important in this process. Their injury has led to the production of tissue factor and tissue factor is released into our forming hemostatic plug. That tissue factor in association with the phospholipid that's being expressed on the platelet surface will lead to the generation of thrombin. So we've gone from 1 to 2 to 3 on your slide. We are going to have then, after thrombin generation, the breakdown, the polymerisation of fibrin. So we're going to break fibrinogen into fibrin fragments that will then polymerize and this will be the more that will cement together the platelet bricks and give us our definitive hemostatic plug, the secondary hemostatic plug. And in fact, this can completely block the lumen and will very definitively stop the bleeding. Even as this process is ongoing, we are trapping up other inflammatory cells and you see that we've trapped red cells and we've trapped neutrophils in this growing hemostatic plug. Those neutrophils will also start part of the antithrombotic counterregulation, they will release proteases that can begin to breakdown the thrombus. 06:02 So again, one of the universal truths in the biologic system, the beauty of the biologic system of the body is that for every positive thing such as thrombosis we have a negative thing and antithrombosis. And this trapping the neutrophils is just the beginning, but endothelial cells again coming into the game are going to express a number of factors that are going to be counterregulatory to the thrombotic process. The endothelial cells will express tissue plasminogen activator, tPA. That will cause the breakdown of that polymerized fibrin, endothelial cells are also going to make thrombomodulin and that will block an early step in the coagulation cascade. So we're already beginning to limit the degree of the thrombosis even as we're thrombosing an injured vessel.
About the Lecture
The lecture Normal Hemostasis by Richard Mitchell, MD, PhD is from the course Hemostasis.
Included Quiz Questions
What covers the basement membrane of a blood vessel?
- Endothelial cell
- Smooth muscle
- Collagen
- Platelets
- Fibrin
An important mediator of primary hemostasis is...
- ...von Willebrand factor.
- ...the tissue factor.
- ...phospholipid complex.
- ...thrombomodulin.
- ...tissue plasminogen activator.
What stops bleeding completely?
- Secondary hemostatic plug
- Antithrombotic counter-regulation
- Thrombin generation
- Platelet adhesion
- Reflex vasoconstriction
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |