Playlist
Show Playlist
Hide Playlist
Aromatic Compounds and Eas: Introduction
-
Slides 06 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
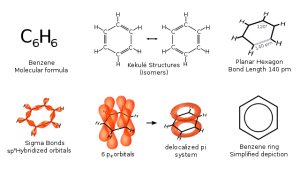
00:01 Right. Okay. So, let’s talk about aromatic ring systems. Now, ostensibly they may look like alkanes; they all contain carbon and hydrogen. But, if we take a look, for example, at the first molecule, benzene, C6H6, you can see that we actually have three double bonds inside. And what we are going to cover in this lecture is what that means in terms of the reactivity of the species and also some of its other properties. 00:28 Benzene derivatives are often shown as the second drawing in the board slide section that you can see here where we have an R group substituting on a benzene ring, where R can either be alkyl or indeed, it can be X, a heteroatom. 00:44 I have also drawn naphthalene and antracene which contain two aromatic rings and three aromatic rings respectively. I have only restricted myself to these three as relatively simple examples. But, you can increase the number of rings and then you start using… looking at large polyaromatic compounds like pi rings. 01:06 These aromatic hydrocarbons, as opposed to the aliphatic ones that we have covered before ,such as cyclohexyl, cyclopentyl groups, are also known as arenes and are all derivatives of that single benzene ring system. In general, aromatic groups can be called aryl groups and are often abbreviated with the letter Ar standing for aryl which means benzene or a related family member. 01:32 So, let’s have a quick look at nomenclature before we actually start looking at the molecular orbitals and how they distinctly influence the reactivity of the benzene ring and make it quite so different from a conventional alkene or indeed, an alkane. Here we have a look at the first example which is ethylbenzene. 01:52 This is where we have monosubstitution. So, we have a single substituent on the benzene ring. In this case, the benzene will go last and whatever our substituent is will go first. 02:03 We have chosen a simple example here where CH2CH3, if you recall, correlates to ethyl derived from ethane. This is ethyl benzene, but equally if it was another carbon in length would be propylbenzene and so on and so forth. Indeed, if you have heteroatom on there, so, for example, a chlorine, it would be chlorobenzene. 02:27 That’s relatively straight forward in the case of monosubstituted benzenes. But, in biology and also in medicine, these are relatively rare as drug treatments. More often than not, so, for example, in the case of paracetamol or as known in American, Acetaminophen, you will find a dye substituted system. 02:47 And this is where we move on to the nomenclature associated with the second example compound. 02:52 Here, we have a benzene ring in the center, as you can clearly identify, with a methyl group in one position and then an amine group, that is a nitrogen bound two hydrogens in a position which is two carbons away. This is known as 3-methyl-aminobenzene. Note in this particular case, as we saw before, when you attach a heteroatom to a carbon and when that heteroatom has a large atomic mass, okay, bearing in mind, nitrogen has an atomic mass of 14 whereas carbon only has an atomic mass of 12, it means that the carbon to which the heteroatom was attached takes priority. 03:33 Therefore, the numbering system for the benzene ring becomes automatic. It travels in a clockwise direction from the amine ring. Obviously, of course, you could flip it on its side and this may effectively mean that the CH3 group would actually be in the five position. But, conventions dictates that you use the shortest distance to get to the other substituent. 03:54 So, in this scenario, we have an aminobenzene with a methyl group in a what is the 3 position with respect to the amine and H2. So, this, therefore, becomes 3-methyl-aminobenzene. 04:08 Now, that’s relatively straight forward when we actually are treating the substituents as the… as those parts which are attached to the benzene ring. However, if more substituents are present in their second example here, their relative position has to be indicated by numbers. If we actually look, however, as benzene as a substituent it is sometimes referred to as a phenyl group. 04:34 This group is sometimes abbreviated Ph, for phenyl, and this is where the name for the third compound here, phenylcyclopentane, comes into place. In this particular case, the core structure is the cyclopentane and we refer to the phenyl group being substituted on that cyclopentane ring, this cycloaliphatic ring that we see here.
About the Lecture
The lecture Aromatic Compounds and Eas: Introduction by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
What is the correct IUPAC name of paracetamol?
- N-(4-hydroxyphenyl)ethanamide
- Acetaminophen
- N- Acetyl-para-aminophenol
- APAP
- Panadol
Which of the following is NOT an aromatic compound?
- 1,2-dimethylcyclopentane
- Anthracene
- 1,2-dibromo-1-phenylcyclooctane
- 1,3,5-trichlorobenzene
- 1,4,5-trimethylphenanthrene
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |