Playlist
Show Playlist
Hide Playlist
Type III Hypersensitivity Reactions
-
Slides Immune-mediated Diseases Type III.pdf
-
Download Lecture Overview
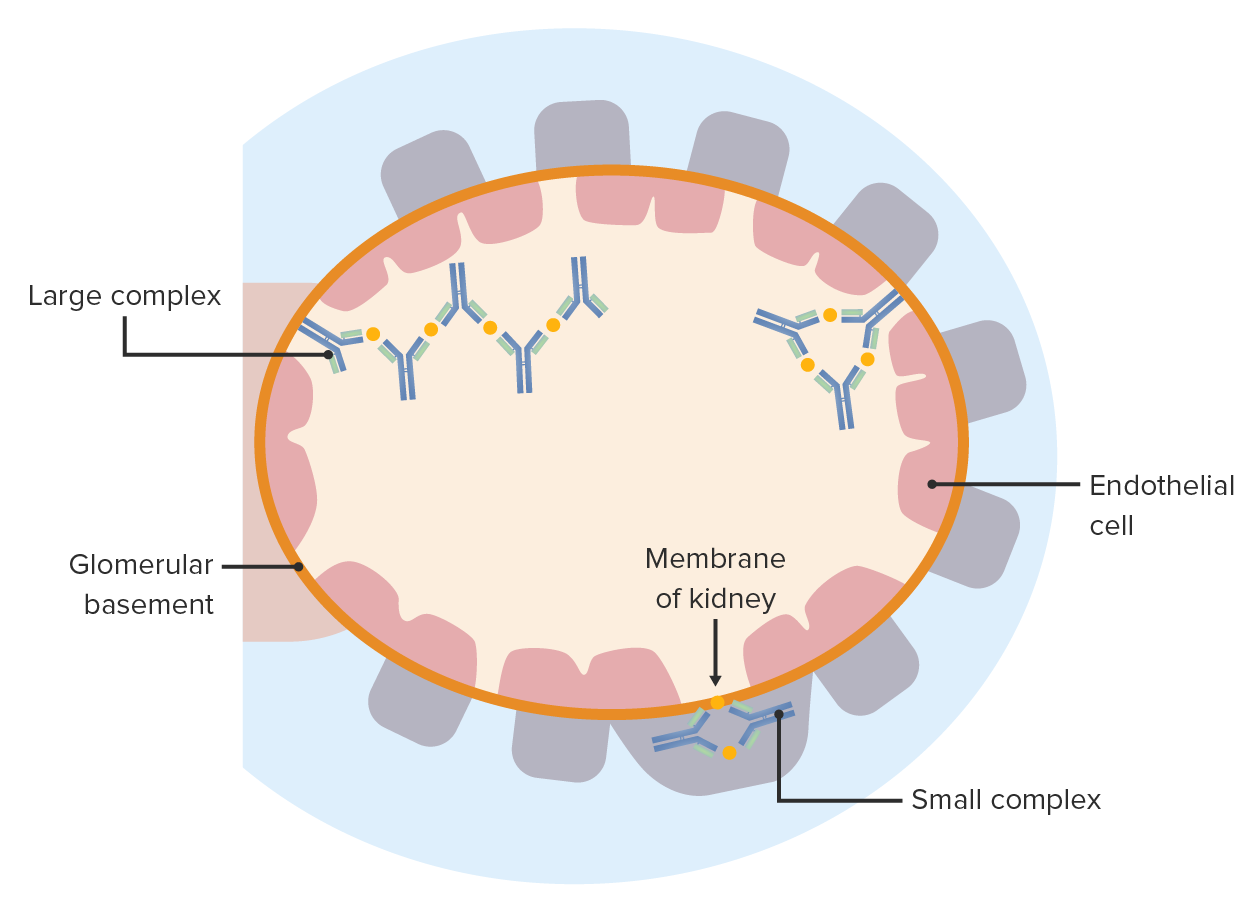
00:01 Hi, welcome back. 00:03 We've talked about type one hypersensitivity, that's due to immunoglobulin isotype II, binding to mast cells, and being crosslinked. 00:11 We talked about type two, that's antibody against fixed tissue antigen. 00:17 At the end of the previous talk on type two, we also talked a little bit about the how immune complexes can occur and how they drive disease. 00:26 And that's we're going to talk about in this session, it's type three hypersensitivity due to IG, antibody antigen antibody immune complex formation. 00:36 So let's think about this in a little bit of detail. 00:38 Again, once we've deposited antibodies that have been bound to an antigen, we can predict what are going to be the mediators, we've already developed the toolbox. 00:50 So a lot of this is going to be hopefully straightforward. 00:53 So we are always generating circulating immune complexes, that is antibody that binds to an antigen, we get multiple kinds of proteins interacting, and most of us, we clear those quite effectively, mainly in the spleen and in the liver through the activity of phagocytic cells. 01:12 But every now and then, you can get circulating immune complexes that can deposit in the vessel wall. 01:17 Again, they're circulating, so where immune complex deposition is going occurs is going to be in vascular beds. 01:23 So once we have that, predictably, we're going to get some consequences of that. 01:30 So we're going to activate complement, and we're going to recruit FC receptor bearing cells to the site where the antigen antibody immune complexes have deposited. 01:40 Those neutrophils are going to come in, they're not going to be able to eat all of the immune complex and they're going to end up dumping their contents there proteases and their arachidonic acid metabolites, all the things that neutrophils will do on to that tissue, which will cause vascular damage. 01:59 And when vascular damage occurs, we're going to get thrombosis, we'll have a schemia in that vascular bed. 02:06 And eventually we'll develop scarring. 02:07 These are all predictable consequences once the immune complexes deposit. 02:14 This is just showing you how the neutrophil granules are being released. 02:17 And there are reactive oxygen species, the Ross remember of superoxide and all that stuff, that's going to cause tissue damage. 02:25 We recognise this as vasculitis, an inflammation and itis of the vessels. 02:32 So, I already told you, most of the time the immune complexes don't deposit, they circulate, we pull them out very effectively by macrophages in the liver and the spleen, and we're good to go. 02:44 But sometimes they do deposit and we'll talk about that. 02:47 So why might they deposit? Well, sometimes it's the size. 02:51 And remember in the previous session, we are talking about giving someone a whole bunch of horse serum albumin and watching them form immune complexes. 03:00 Smaller immune complexes are going to be harder to clear because they have less FC, and they're going to be much more likely to deposit. 03:07 So bigger complexes tend to get cleared more effectively, smaller complexes not so much. 03:14 It's also going to be the physical chemical characteristics. 03:17 So charge, how effectively the antibody is bound. 03:22 The kind of three dimensional conformation of the antigen antibody complex will deposit, will determine whether it deposits or not. 03:32 If the macrophages in the spleen, in in the liver or otherwise compromised, they are sopping up lipid or something else, you may not have adequate phagocyte clearance capacity. 03:45 And then those circulating immune complexes will be able to go around much more within the circulation and eventually will deposit. 03:53 Hemodynamics plays a big role in this. 03:56 So, smaller vascular beds where there's more sluggish flow such as joints and the kidney nephron are going to be favoured targets for the deposition of immune complexes. 04:08 The endothelial characteristics are also going to be very important. 04:11 Remember that the endothelium in the renal glomerulus is a fenestrated endothelium, it's got holes in it. 04:18 So it's possible are much easier in that environment for immune complexes to get deposited in the basement membrane. 04:26 It will also depend to some extent on vascular permeability. 04:29 So if we make the vessels leaky because of injury, or because cytokines or because of an allergy or mast cell degranulation, all those things will potentially open up gaps in the microvasculature where the immune complexes can deposit and also, if we are activating neutrophils for any variety of reasons, and they are causing local damage that will also create an opportunity and environment by which the immune complexes can be deposited. 05:00 So all these things will potentially impact whether or not you get immune complex deposition.
About the Lecture
The lecture Type III Hypersensitivity Reactions by Richard Mitchell, MD, PhD is from the course Immune-mediated Diseases.
Included Quiz Questions
Which of the following factors does not play a role in the deposition of immune complexes in a type III hypersensitivity reaction?
- Protein content of the immune complex
- Size of the immune complex
- Charge on the immune complex
- Rate of production of the immune complex
- Rate of clearance of the immune complex
In a type III hypersensitivity reaction, where are immune complexes typically deposited?
- Basement membrane
- Endothelium
- Mesothelium
- Epithelium
- Lamina propria
The pathogenesis of a type III hypersensitivity reaction involves...
- ...the deposition of immune complexes in tissues and small blood vessels.
- ...autophagy.
- ...the release of vasoactive amines and cytokines from mast cells.
- ...cell-mediated cytotoxicity.
- ...antibody-dependent cellular cytotoxicity.
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |