Playlist
Show Playlist
Hide Playlist
Fluid Replacement Therapy (FTP) in Children: Diagnosis
-
Slides IVfluids Pediatrics.pdf
-
Download Lecture Overview
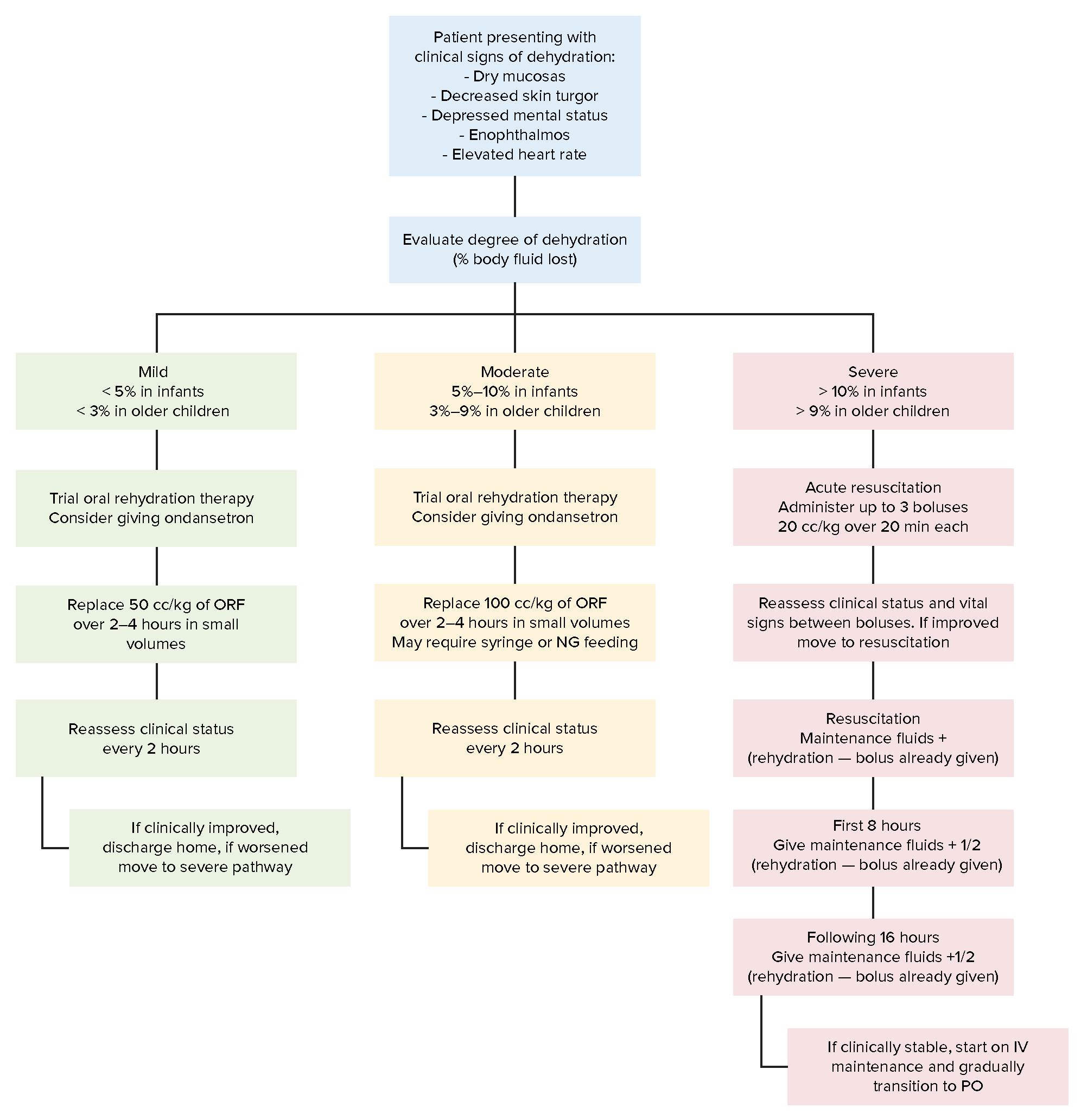
00:01 So if we see a child with dehydration, it’s indicated to get a Chem-7, especially for severe dehydration because we’re concerned about imbalance of electrolytes. 00:15 And the electrolyte that gets children in trouble the most is actually sodium. 00:21 It is not necessary to get a Chem-7 for a child who has mild dehydration unless you have other concerns about the child. 00:29 Because those children, once their fluids have been replenished, will normalize their Chem-7. 00:35 So this is how a Chem-7 on your slide here is organized when we draw it out shorthand on a piece of paper. 00:42 Sodium, potassium, chloride, bicarb, BUN and creatinine, and glucose. 00:47 Let’s go through some examples. 00:50 Here are normal values for your Chem-7. 00:53 The sodium is 140, the potassium is around 3.8. 00:57 In this child, the chloride is normal at 110, and a normal bicarb is 24. 01:02 That 24 and that 140 are usually fairly carefully constricted at that normal value. 01:10 The next numbers, the 5 and 0.2, are the BUN and creatinine. 01:13 Those will herald signs of dehydration if the BUN is very high. 01:18 But the creatinine will remain normal unless the child is in renal failure or very severely dehydrated. 01:25 Glucose may be not so important in adults, but in children, it’s critical. 01:30 Because young children, when they get dehydrated, often get hypoglycemic. 01:35 This is because children’s liver stores of glycogen are much smaller than adults. 01:42 That means that without being fed for a period of time, they’re at much more risk for hypoglycemia and potentially even seizure from a prolonged period of dehydration. 01:54 So here’s what a mild dehydration may look like. 01:58 You can see our patient’s sodium, potassium, chloride and bicarb are all doing okay. 02:04 The BUN has started coming up a little bit and you can see the glucose has come down to 70, starting to drift down. 02:11 You might even see a slightly low bicarbonate, 19 or 20 wouldn’t be surprising. 02:17 But those are the key things that you’re going to see first. 02:20 Here’s a child with moderate dehydration. 02:23 The sodium is starting to creep up. 02:25 In this case, this child is just not drinking. 02:28 If the child were eating, say some liquid, but losing more liquid than she were drinking, that child might have a low sodium even in the face of dehydration. 02:40 You can see that now, the child’s bicarbonate is low. 02:45 It’s significantly acidotic all the way down to 13. 02:49 And the chloride is starting to creep up as a compensatory method. 02:54 But there’s something special about this that we have to note and it has to do with the sodium, the chloride and the bicarb. 03:01 Remember your equation for calculating a gap. 03:05 In this case, this child has large anion gap, which is a sign that this child is also having a metabolic acidosis. 03:14 I’m guessing this child either has ketones or this child has lactic acidosis, which might be driving that gap. 03:22 Again, the BUN is coming up and now the creatinine is coming up and look how low that glucose is getting. 03:27 Not surprising at all for a child with moderate dehydration. 03:31 So all these numbers are starting to become pretty significant and the anion gap is 20, 145 minus 112 minus 13. 03:42 In severe dehydration, now we’re really getting into trouble. 03:46 This child’s sodium is still high. 03:48 It might get much higher and we have to do some very fancy calculations to fix. 03:53 But of note, the chloride’s coming up. 03:56 The bicarb’s coming way down and the BUN and creatinine are significantly elevated. 04:00 Look how hypoglycemic this child is. 04:02 You can see how this will happen
About the Lecture
The lecture Fluid Replacement Therapy (FTP) in Children: Diagnosis by Brian Alverson, MD is from the course Pediatric Gastroenterology.
Included Quiz Questions
What lab anomaly are children with severe dehydration at risk for that adults are less likely?
- Hypoglycemia
- Transaminitis
- Metabolic alkalosis
- Hypokalemia
- Hyperkalemia
Which of the following combination is known as Chem 7?
- Sodium, potassium, bicarbonate, chloride, BUN, creatinine, and glucose
- Sodium, potassium, bicarbonate, chloride, BUN, creatinine, and pH
- Sodium, phosphate, bicarbonate, chloride, BUN, creatinine, and glucose
- Sodium, potassium, bicarbonate, magnessium, BUN, creatinine, and glucose
- Sodium, potassium, bicarbonate, chloride, urea, creatinine, and glucose
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Very clear explanation of how the lab values change according to the level of dehydration.