Playlist
Show Playlist
Hide Playlist
Pathogenesis – TSEs
-
Slides 15 Prions MicrobiologyAdvanced.pdf
-
Download Lecture Overview
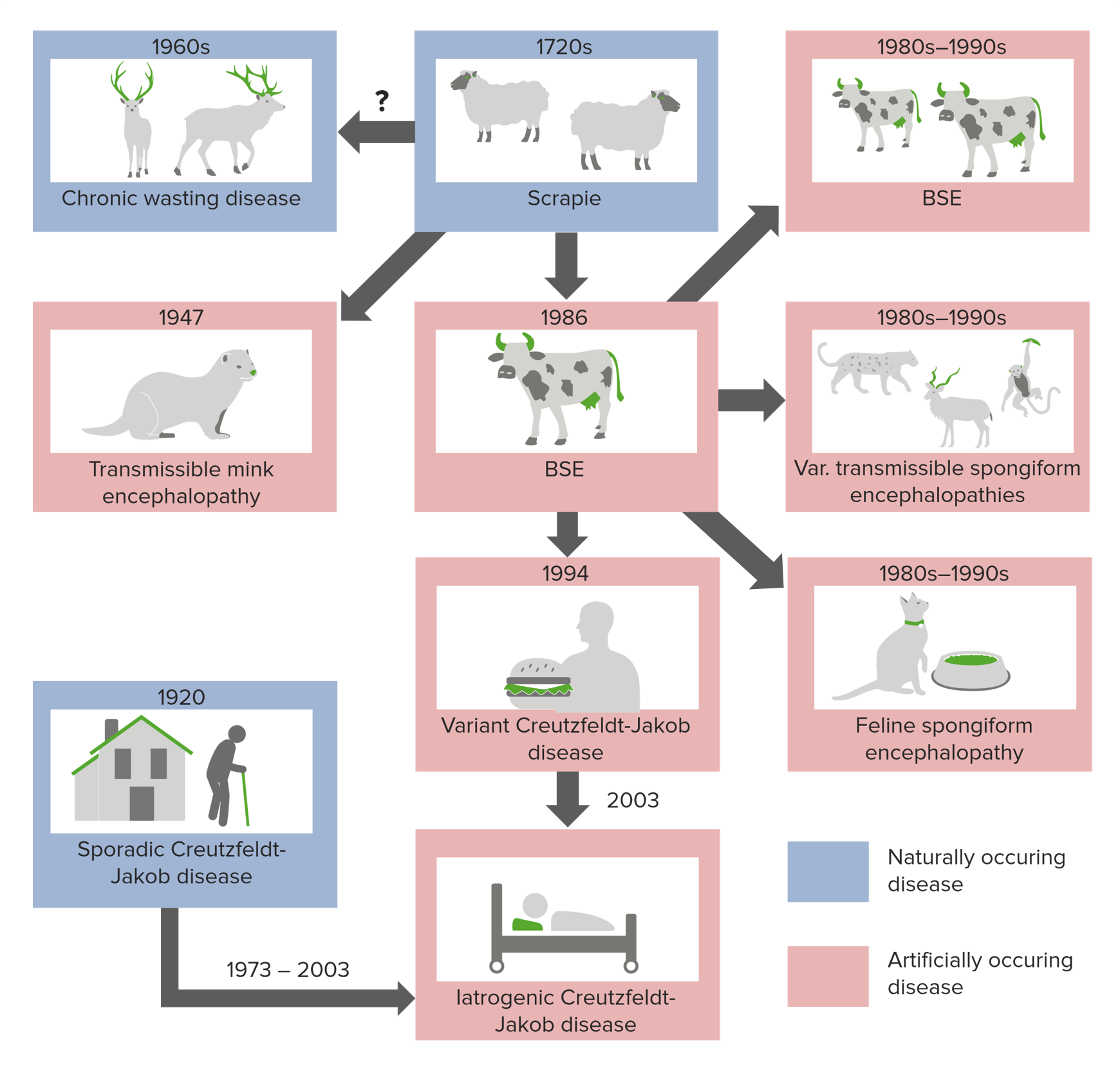
00:01 When you acquire a misfolded protein, the agent first accumulates in the lymphoreticular and secretory organs, and eventually spreads to the central nervous system. And in the central nervous system, the pathology includes death of astrocytes, astrocytosis, vacuolization, the holes in the central nervous system, the brain that makes it be called spongiform TSE, that's the 'S' in TSE. And you eventually lose neurons, slowly but surely neurons go, and you lose many of your functions. You have cerebellar ataxia. What does that mean? You can't walk, you have more and more trouble walking, until at one point you can no longer walk. You tend to have dementia, you go crazy, you don’t recognize people, you start to just stare and you stop talking, akinetic mutism, you simply stare straight ahead without saying a word. There are other issues too, you can have psychological problems, you can yell and scream at people for no reason, depends on which TSE you are getting. And then in any time from months to years, it can be a year or it can be 20 years of incubation, you die. As I said, these are uniformly fatal, no way to prevent the death. So pretty scary and a bit insidious. They come from nowhere. There's no inflammatory antibody or cellular response to this protein. It's a normal cell protein. It's just misfolded and our immune system does not recognize it as different, so you don't make any antibody response, so there is no way that you yourself can prevent this disease from happening. Only if we can somehow give you some drugs one day, which we don't yet have. 01:42 Alright, I’m going to tell you about one test for TSEs that was developed a long time ago, which for many years was the only way you can tell postmortem, whether someone had a TSE. And I said postmortem because you need a little brain tissue in order to do this, and when someone presents their doctor with early symptoms of a TSE, there are in no shape to have a brain biopsy, so this is done after they die. The observation was that PrPc, the normal protein, we see it in its structure on the right, on the left is a stretched out version of the protein. If you take say a brain extract from someone who died of a TSE and you digest it with an enzyme that digests proteins called proteinase K. That enzyme will completely digest this protein away into bits, and if you run a western blot, which is a way to detect PrPc specifically with an antibody, you won’t see anything because it's digested away. If you take the PrPsc version, which now has lots of beta sheets and you do the same thing, you digest it with proteinase K, that is largely resistant to digestion. 02:53 You clip off a little bit at the left end of the protein, the end terminus, but pretty much the protein is resistant to proteinase K digestion. So right away this is a nice way to distinguish the normal protein from the pathogenic protein. And you could take a sample from a person and subject it to this assay. If all the PrP is digested, they don’t have a TSE, but if it's resistant, they do have a TSE. And you may ask, what does it matter after they die? Well you want to know what people died of, and you want to make sure they're not going to contaminate someone else. So for example very recently, someone showed up at an emergency room, they had shot themself in the head, an attempted suicide, and only after few hours did they find out that this person had a TSE, he was going to die, so they decided to take their life, but in the meantime he contaminated all the emergency room with brain material and if they didn't realize he had a TSE, which they figured out by a diagnostic test, lots of other people could've acquired it as well.
About the Lecture
The lecture Pathogenesis – TSEs by Vincent Racaniello, PhD is from the course Prions.
Included Quiz Questions
Which organ is most affected by prion diseases?
- Brain
- Lungs
- Kidneys
- Heart
- Appendix
After treatment with proteinase K, which laboratory method can be used to identify abnormal prion proteins?
- Western blot
- Microscopic examination
- Culture on Sabouraud agar
- UV rays
- FNA
Which of the following signs, symptoms, or lab findings is NOT found in transmissible spongiform encephalopathies?
- Wartenberg's sign
- Death of astrocytes
- Vacuolization
- Ataxia
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |