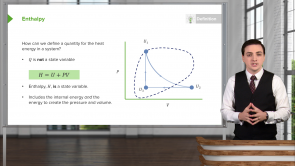
Pressure-Volume Diagrams

About the Lecture
The lecture Pressure-Volume Diagrams by Jared Rovny, PhD is from the course Thermodynamics and Thermochemistry.
Included Quiz Questions
What is the formula for the amount of work done by the system at constant pressure?
- Amount of work done = pressure x change in volume
- Amount of work done = pressure x change in energy
- Amount of work done = pressure x change in temperature
- Amount of work done = pressure x change in the length of the system
- Amount of work done = change in internal energy
System A undergoes an adiabatic process from volume V₁ and pressure P to volume V₂ > V₁. System B undergoes an isothermal process from volume V₁ and pressure P to volume V₂ > V₁. Which system has done more work and why?
- System B has done more work because in the pressure-volume diagram the curve of system B remains above the curve of system A throughout the process.
- System B has done more work because in the pressure-volume diagram the curve of system B remains below the curve of system A throughout the process.
- System A has done more work because in the pressure-volume diagram the curve of system A remains above the curve of system B throughout the process.
- System A has done more work because in the pressure-volume diagram the curve of system A remains below the curve of system B throughout the process.
- Both have done the same amount of work since the area under their curves in the pressure-volume diagram is the same.
When is a system considered isochoric, isothermal, adiabatic or isobaric?
- Isochoric : volume held constant, isothermal: temperature held constant adiabatic: No heat transfer (Q = 0), isobaric: pressure held constant
- Isochoric : volume held constant, isothermal: No heat transfer (Q = 0) adiabatic: temperature held constant, isobaric: pressure held constant
- Isochoric : pressure held constant, isothermal: temperature held constant adiabatic: No heat transfer (Q = 0), isobaric: volume held constant
- Isochoric : volume held constant, isothermal: temperature held constant adiabatic: pressure held constant, isobaric: No heat transfer (Q = 0)
- Isochoric : volume held constant, isothermal: pressure held constant adiabatic: No heat transfer (Q = 0), isobaric: temperature held constant
The volume of a gas is doubled as its pressure is held constant at 1 atm. If the initial volume of the gas is 1 L, how much work is done by the gas? (1 atm = 100,000 Pa, 1 L = 0.001 m³)
- 100 J
- 200 J
- 50 J
- -200 J
- -100 J
These courses may be of interest to you
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |