Gibbs Free Energy

About the Lecture
The lecture Gibbs Free Energy by Jared Rovny, PhD is from the course Thermodynamics and Thermochemistry.
Included Quiz Questions
What is the formula for the change in Gibbs free energy at constant temperature?
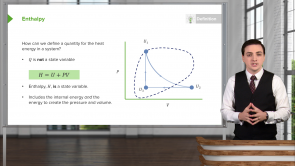
- ΔG = ΔH - TΔS
- ΔG = ΔG = ΔH/T - ΔS
- ΔG = ΔS - ΔH/T
- ΔG = ΔH + TΔS
- ΔG = ΔH/T + ΔS
Which of the following change in Gibbs free energies represents a spontaneous reaction?
- ΔG = -10 kJ
- ΔG = 0
- ΔG = 10 kJ
- ΔG = 100 kJ
- ΔG = 50 kJ
Which of the following is favorable for a reaction?
- Minimize energy and maximize entropy
- Maximize energy and maximize entropy
- Minimize energy and minimize entropy
- Minimize entropy and maximize energy
- Maximize entropy and maximize enthalpy
What is ΔG for a reaction if ΔH is equal to 36 kJ, ΔS is equal to 125 J/K and the reaction occurs at 298 K? Is this reaction spontaneous?
- ΔG = -1.25 kJ < 0, hence the reaction is spontaneous
- ΔG = 1.25 kJ > 0, hence the reaction is not spontaneous
- ΔG = 0, hence the reaction is in equilibrium
- ΔG = -1.25 kJ < 0, hence the reaction is not spontaneous
- ΔG = 1.25 kJ > 0, hence the reaction is spontaneous
These courses may be of interest to you
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |